Laboratory Activity 1
Expansion by Heat
I. Materials:
a large bottle with a narrow neck
a large container for hot water
inflated balloon
II. Diagrams/Pictorials
a large bottle with a narrow neck
a large container for hot water
inflated balloon
II. Diagrams/Pictorials
III. Results and Discussion:
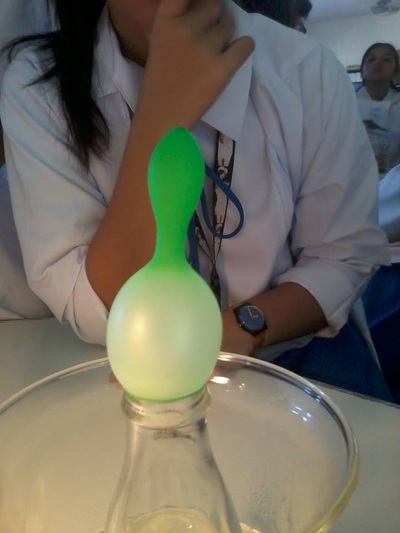
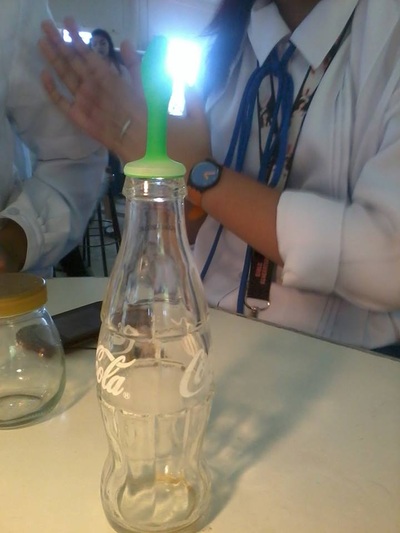
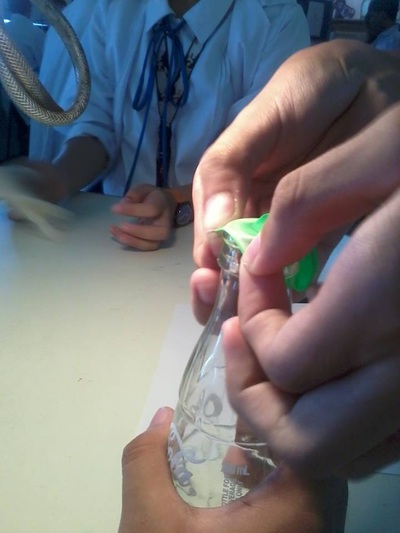
Based on the experiment, the balloon expanded when the bottle was immersed in the hot water. On the other hand, it contracted when the bottle was placed on top of the table. It was observed that the amount of air rose and increased when the bottle was immersed due to the hot air molecules. On the contrary, the amount of air went down and decreased when it was place on the table causing the balloon to shrink.
IV. Conclusion:
The higher the temperature, the higher the amount of volume. When a body of object is heated, it expands. On the contrary, when a body of object is cooled, it contracts. This explains why temperature affects the amount of volume. Therefore, the temperature is directly proportional to volume or vice versa.
V. Source:
i-Physics IV, Heat and Temperature
Based on the experiment, the balloon expanded when the bottle was immersed in the hot water. On the other hand, it contracted when the bottle was placed on top of the table. It was observed that the amount of air rose and increased when the bottle was immersed due to the hot air molecules. On the contrary, the amount of air went down and decreased when it was place on the table causing the balloon to shrink.
IV. Conclusion:
The higher the temperature, the higher the amount of volume. When a body of object is heated, it expands. On the contrary, when a body of object is cooled, it contracts. This explains why temperature affects the amount of volume. Therefore, the temperature is directly proportional to volume or vice versa.
V. Source:
i-Physics IV, Heat and Temperature